 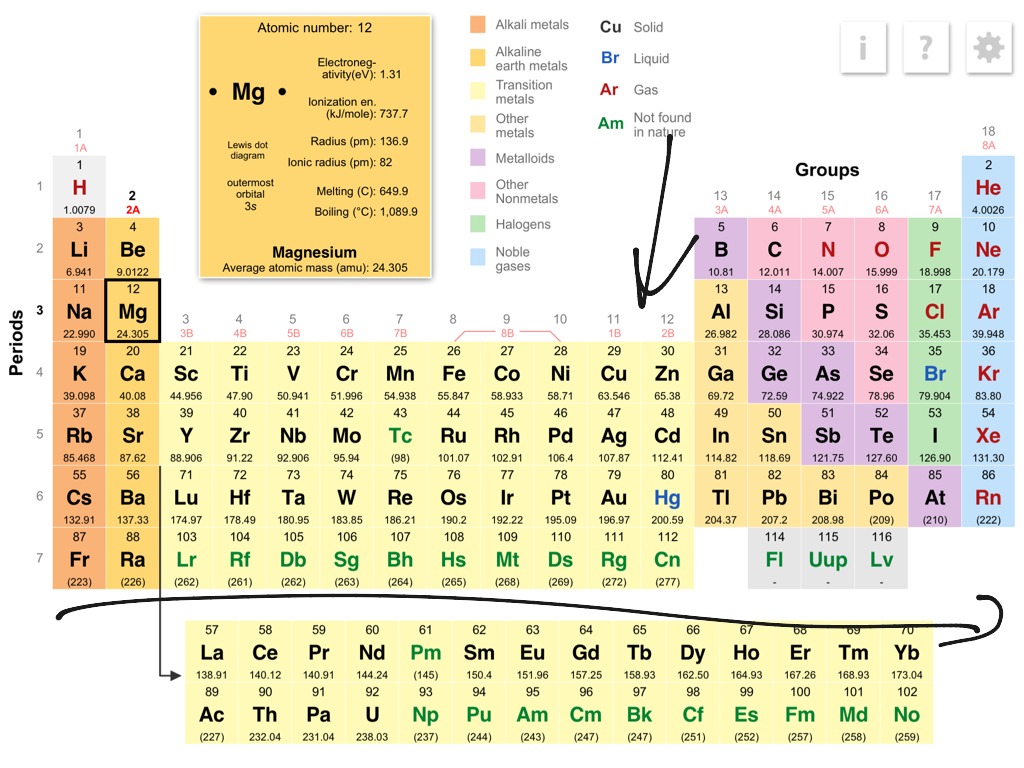 
In the modern IUPAC nomenclature, the alkali metals comprise the group 1 elements, excluding hydrogen (H), which is only nominally considered a group 1 element. Because of their high reactivity, they must be stored under oil to prevent reaction with air, and are found naturally only in salts and never as the free element. They can all be cut easily with a knife due to their softness, exposing a shiny surface that tarnishes rapidly in air due to oxidation by atmospheric moisture and oxygen. Cesium is one of the few elemental metals that is liquid at room temperature, but also reacts explosively with water. The alkali metals have very similar properties: they are all shiny, soft, highly reactive metals at standard temperature and pressure and readily lose their outermost electron to form cations with charge +1. Therefore, for practical purposes, Cesium is often regarded as the most reactive metal. The alkali metals provide the best example of group trends in properties in the periodic table, with elements exhibiting well-characterized homologous behaviour. The alkali metals have their outermost electron in an s-orbital and this electronic configuration results in their characteristic properties. The structures of the metals can be summarised by the table below which shows that most metals crystallise in roughly equal amounts of bcc, hcp and ccp lattices.Ĭrystal structure of metallic elements in the periodic table Mercury, caesium and gallium have melting points below 30 ☌ whereas all the other metals have sufficiently high melting points to be solids at "room temperature". For a given period, the melting points reach a maximum at around Group 6 and then fall with increasing atomic number. In the alkali metals (Group 1) and alkaline earth metals (Group 2) the melting point decreases as atomic number increases, but in transition metal groups with incomplete d-orbital subshells, the heavier elements have higher melting points. The transition metals have generally higher melting points than the others. Melting points are chosen as a simple measure of the stability or strength of the metallic lattice. cubic close packed (face centered cubic) has every third layer directly above/below each other, A,B,C,A,B,C.hexagonal close packing has alternate layers positioned directly above/below each other, A,B,A,B. 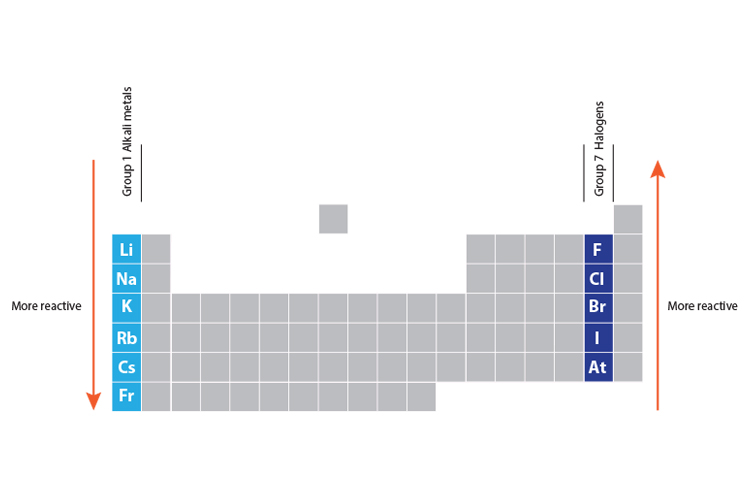
It can be envisaged that for a regular buildup of layers: If the close packed structures are considered as being built of layers of spheres then the difference between hexagonal close packing and cubic close packed is how each layer is positioned relative to others. For closest packing, every atom has 12 equidistant nearest neighbours, and therefore a coordination number of 12. A simple model for both of these is to assume that the metal atoms are spherical and are packed together in the most efficient way (close packing or closest packing). cubic close packed (face centred cubic) and hexagonal close packed structures. Many metals adopt close packed structures i.e. This basic repeating unit is called a unit cell. \)Ĭrystalline solids consist of repeating patterns of its components in three dimensions (a crystal lattice) and can be represented by drawing the structure of the smallest identical units that, when stacked together, form the crystal. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
